Hydroxychloroquine
Hydroxychloroquine is a highly applicable drug processed out of extracts from the bark of the tropical Cinchona tree. Hydroxychloroquine is one of the 4-aminoquinoline medicines.
History
Medical Uses
Hydroxychloroquine is one of the most repurposed drugs in the history of medicine.
- Hydroxychloroquine as Antimalarial
- Hydroxychloroquine as Antiviral including SARS-CoV-2 infection and COVID-19 disease
- Hydroxychloroquine as treatment for autoimmune disorders
Coronavirus
During the COVID-19 pandemic, hydroxychloroquine become controversial.
Prior to the pandemic, there was a substantial body of literature suggesting that hydroxychloroquine would most likely help stave off a SARS-CoV epidemic. The following paragraphs are taken from a Rounding the Earth article.1)
In late 2004, in the wake of the SARS-CoV outbreak that claimed over 8,000 lives across nearly 30 nations, Belgian researcher Els Keyaerts and her team published a paper in the journal Biochemical and Biophysical Research Communications entitled “In vitro Inhibition of Severe Acute Respiratory Syndrome Coronavirus by Chloroquine”.2) Keyaerts and her cohorts tested the antiviral properties of chloroquine phosphate on a line of kidney cells extracted from an African green monkey. Using amounts of CQ similar to those used to treat malaria patients, the researchers noted successful antiviral effects (killing of the virus) and cytostatic effects (inhibition of viral replication). The researchers concluded, “Chloroquine, an old antimalarial drug, may be considered for immediate use in the prevention and treatment of SARS-CoV infections.”
Keyaerts’s paper was not the first to explore the effects of CQ and other quinolines on viruses. For decades, scientists have found CQ to be effective in vitro, in vivo, or in humans against flaviviruses such as dengue virus, retroviruses such as HIV, myxovirus, Zika virus, and picornaviruses such as poliovirus. And it does this while working against cell damage caused by some viruses. Less than a year after Keyaerts et al published, an American research team led by Martin J. Vincent published a study in Virology Journal titled “Chloroquine is a potent inhibitor of SARS coronavirus infection and spread,” reporting in vitro viral inhibition both as prophylaxis and treatment. From that research:
In addition to the well-known functions of chloroquine such as elevations of endosomal pH, the drug appears to interfere with terminal glycosylation of the cellular receptor, angiotensin-converting enzyme 2. This may negatively influence the virus-receptor binding and abrogate the infection, with further ramifications by the elevation of vesicular pH, resulting in the inhibition of infection and spread of SARS CoV at clinically admissible concentrations.
This interference with the ACE2 receptor adds to the rationale for testing HCQ efficacy on those infected with SARS-CoV-2, and COVID-19 patients.
This level of versatility of HCQ is quite unusual among drugs. Few drugs are successfully repurposed, so we might wonder: What is it about HCQ that makes it so useful? While nobody knows that answer completely, at least some of HCQ’s medicinal potential stems from a wide array of effects. First, for reasons researchers do not fully understand, HCQ and CQ block the parasites that cause most malaria from entering the blood stream via the CD147 pathway, which is noteworthy because CD147 is one of the cellular pathways through which SARS-CoV-2 passes (into the bloodstream where it can cause the debilitating microthrombotic effects associated with COVID-19).
In 2007, a team of French infectious disease experts experienced with these medications reviewed the literature, noting the rise of coronavirus epidemics, and predicted that HCQ and CQ would prove even more widely applicable to new diseases through the twenty-first century.
It is clear that for many years, scientists focused on HCQ as a drug with potential to shut down the next coronavirus outbreak. Many rationale papers and opinion pieces were published in medical journals, and a few even in blogs, both years before the pandemic and during its early months. At the onset of the SARS-CoV-2 pandemic, hundreds of researchers quickly began registering trials to study the effects of HCQ or CQ both in the U.S. and around the world. In fact, through April 27, 2020, 46 of the 68 repurposed medicine clinical trials registered for prophylaxis (68%) proposed testing HCQ out of all possible options!
Clearly, HCQ was broadly considered the obvious first candidate among researchers as a drug to treat the next coronavirus epidemic (even if our media didn't want to speak a word about it).
There was one single rationale paper for repurposing medicine published during the early going of the pandemic that did not include any discussion at all of hydroxychloroquine, despite its being the obvious first choice. That's the one that has NIAID Director Dr. Anthony Fauci's name on it.
Hydroxychloroquine blocks SARS-CoV-2 entry into the endocytic pathway in mammalian cell culture
Hydroxychloroquine blocks SARS-CoV-2 entry into the endocytic pathway in mammalian cell culture
Zixuan Yuan, Mahmud Arif Pavel, Hao Wang, Jerome C. Kwachukwu, Sonia Mediouni, Joseph Anthony Jablonski, Kendall W. Nettles, Chakravarthy B. Reddy, Susana T. Valente & Scott B. Hansen
Communications Biology volume 5, Article number: 958 (2022) Cite this article
Abstract
Hydroxychloroquine (HCQ), a drug used to treat lupus and malaria, was proposed as a treatment for SARS-coronavirus-2 (SARS-CoV-2) infection, albeit with controversy. In vitro, HCQ effectively inhibits viral entry, but its use in the clinic has been hampered by conflicting results. A better understanding of HCQ’s mechanism of actions in vitro is needed.
Recently, anesthetics were shown to disrupt ordered clusters of monosialotetrahexosylganglioside1 (GM1) lipid. These same lipid clusters recruit the SARS-CoV-2 surface receptor angiotensin converting enzyme 2 (ACE2) to endocytic lipids, away from phosphatidylinositol 4,5 bisphosphate (PIP2) clusters.
Here we employed super-resolution imaging of cultured mammalian cells (VeroE6, A549, H1793, and HEK293T) to show HCQ directly perturbs clustering of ACE2 receptor with both endocytic lipids and PIP2 clusters. In elevated (high) cholesterol, HCQ moves ACE2 nanoscopic distances away from endocytic lipids. In cells with resting (low) cholesterol, ACE2 primarily associates with PIP2 clusters, and HCQ moves ACE2 away from PIP2 clusters—erythromycin has a similar effect.
We conclude HCQ inhibits viral entry through two distinct mechanisms in high and low tissue cholesterol and does so prior to inhibiting cathepsin-L. HCQ clinical trials and animal studies will need to account for tissue cholesterol levels when evaluating dosing and efficacy. 3)
Pfizer Recomends Hydroxychloroquine for SARS-CoV-2 Treatment
 Pfizer STILL Recommends Hydroxychloroquine for the Treatment of Severe Covid Disease
How can we ever trust SCIENCE again after such a blatant heresy by its flesh-and-blood corporate theophany?
Pfizer STILL Recommends Hydroxychloroquine for the Treatment of Severe Covid Disease
How can we ever trust SCIENCE again after such a blatant heresy by its flesh-and-blood corporate theophany?
Substack - December 13, 2022 by Ashmedai
If you just want to see where Pfizer recommends HCQ and skip the article, it is on page 13 here.4)
I discovered a while back that Pfizer recommends Hydroxychloroquine for the treatment of severe late-stage covid disease.
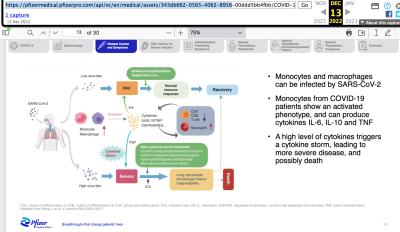
On a whim, I decided to check to see if Pfizer was still recommending HCQ. I suspected that there was a pretty good chance that Pfizer had pulled their prior guidance, because not only should it be a profound embarrassment to be caught advocating for the most salacious and vile lunatic disinformation of the pandemic (besides the anti-covid vaccine claims), Pfizer also now had an added incentive to push people towards Paxlovid.
Initially, it seemed like Pfizer had pulled their little slide deck with the heretical medical advice DISINFORMATION, because when I tried the link I put in the original article above - COVID 19: Clinical Presentation - it led to this - 404 Page Not Found 5)6)
NOTE - Pfizer pdf references at Wayback copies w first link to HCQ diagram above 7)8)
Interactions
Pharmacology
Hydroxychloroquine is a drug with a “Goldilocks zone” dosage level where too little is generally ineffective, but too much can be toxic.
Contraindications
Safety Data
ThermoFisher Scientific safety data sheet for hydroxychloroquine.9)
Adverse Events
Mechanisms
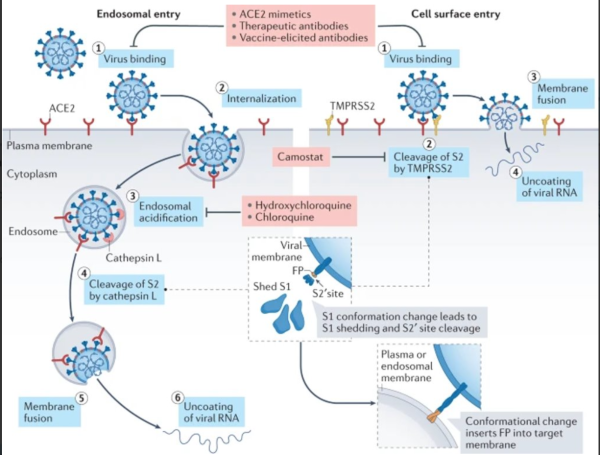 If hydroxychloroquine could help with Omicron, would politics block its use?
Dr Philip McMillan - Oct 31, 2022 10)
If hydroxychloroquine could help with Omicron, would politics block its use?
Dr Philip McMillan - Oct 31, 2022 10)
- Hydroxychloroquine blocks SARS-CoV-2 entry into the endocytic pathway in mammalian cell culture11)

