COVID-19 Vaccine Research and Development
This is a summary page for research and development related to any and all COVID-19 vaccines.
The various vaccines targeting SARS-CoV-2 or COVID-19 have undergone historically rapid development, both in the United States and in other nations. The U.S. federal government's program for rapid COVID-19 vaccine development was dubbed Operation Warp Speed.
History of Coronavirus Vaccine R&D
Prior to the COVID-19 pandemic, no vaccine of any type had proved safe and effective in combatting coronaviruses.
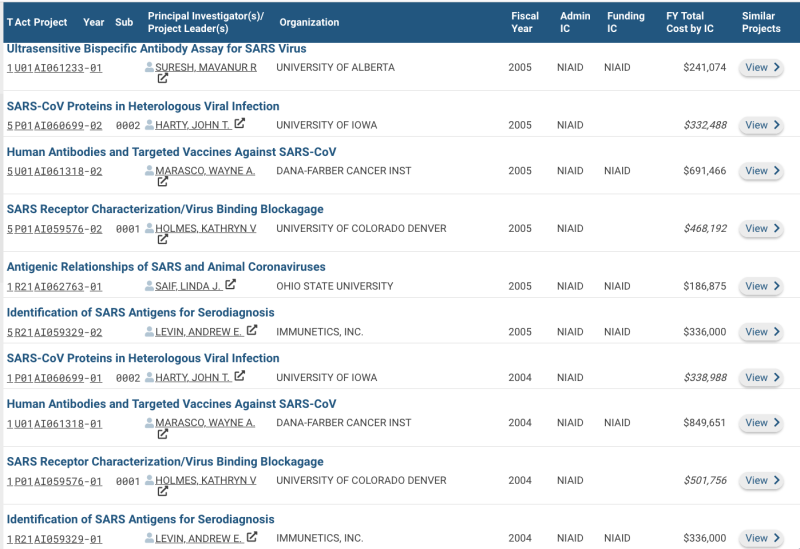
NIH Funding SARS - Coronavirus Vaccine Development
2002
Coronavirus vaccine development Project Number1ZIAAI005125-02 Contact PI/Project LeaderGRAHAM, BARNEY Awardee OrganizationNATIONAL INSTITUTE OF ALLERGY AND INFECTIOUS DISEASES
When the MERS CoV outbreak raised global health concerns, we initiated a program to develop a candidate vaccine. Starting from Spike glycoprotein (S) sequences, we developed an immunization strategy consisting of a full-length S DNA prime and a S1 subunit protein boost that elicited high titers of neutralizing antibodies against eight different MERS-CoV strains. Immune sera contained potent neutralizing antibodies targeting the receptor binding domain (RBD), non-RBD portions of S1, and the S2 subunit.
From the immunized mice we produced a panel of hybridomas and produced monoclonal antibodies from which a variety with high neutralizing activity were selected for further characterization. The atomic structure of a monoclonal antibodyD12in complex with the RBD revealed two distinct mechanisms by which they block binding to the MERS-CoV receptor, DPP4. In addition, immunogenicity was measured in nonhuman primates. Thus, vaccine immunogens designed from S sequences induced a diverse repertoire of neutralizing antibodies, demonstrating an efficient approach to vaccine design that may be applicable to other emerging viruses. Structural studies were also initiated with the spike glycoprotein of the HKU1 beta-coronavirus, to explore structure and for receptor discovery. Human airway epithelial cell culture system was established to initiate entry and pathogenesis studies of HKU1. 1)
2004
Human Antibodies and Targeted Vaccines Against SARS-CoV Project Number5U01AI061318-05 Contact PI/Project Leader MARASCO, WAYNE A. Awardee Organization DANA-FARBER CANCER INSTITUTE
Effective prophylaxis and therapies are urgently needed in the event that there is reemergence of the highly contagious and often lethal severe acute respiratory syndrome (SARS) coronavirus (SARS-CoV) infection. Passive immunization has proven to be an effective and safe strategy for the prevention and treatment of viral diseases. In this proposal we will describe preliminary data in which we have identified a high-affinity human monoclonal antibody termed 80R that acts as a potent SARS-CoV entry inhibitor.
The strategies that were used to identify 80R Mab will now be incorporated in a broad effort, to identify other neutralizing epitopes on the SARS-CoV that can be used to develop human Mabs and vaccine candidates. Our studies will include a systematic effort to explore the mechanism(s) of escape that may develop against the 80R and other neutralizing epitopes of the SARS-CoV and how these escape mechanisms can be thwarted to enable these treatment and vaccine strategies to succeed. Accordingly, the specific aims of this proposal are to establish a high producer cell line and master cell bank of the 80R transfectoma and produce gram quantities of 80R human IgG1 Mab. The Mab will be used to test its efficiency to prevent SARS-CoV infection in an animal model. Our initial studies will be performed initially in a mouse model in which we test the ability of the 80R Mab given pre and post virus inoculation to prevent SARS-CoV infection.
If these studies are successful, we will move into a non-human primate model of SARS to determine if this human Mab can ultimately be used in the emergency prophylaxis and treatment of SARS in humans. We will also study the mechanisms of neutralization escape that may develop after treatment of SARS-CoV with 80R Mab. For these studies, we will generate S1 neutralization escape mutants in vitro and then characterize the S1 mutants for the amino acid changes in the S1 protein, the changes in binding affinity of the mutant S1 proteins for the ACE2 receptor and for 80R Mab, the ability of the S1 mutant viruses to enter cells at lower ACE2 receptor concentrations and the ability of the S1 mutant viruses induce cytopathic and pathogenic effects in vitro and in vivo. We will also identify neutralizing human monoclonal antibodies to epitopes other than the 80R epitope on S1 protein and to isolate high-affinity neutralizing human antibodies against S2 and membrane (M) protein.
We will identify antigenic protein fragments (APFs) from the SARS-CoV Spike (S) and M proteins by screening with natural peptide libraries derived from SARS-CoV. For these studies, we will use the naturally occurring neutralizing antibodies in convalescent SARS serum as well as the 80R Mab and the additional neutralizing antibodies to identify the neutralizing epitopes. Finally, these APFs will be incorporated into the design of optimal forms of S-IgG and M-IgG fusion proteins that can be used as immunogens for induction of humoral immune responses against SARS viral proteins, which, in turn, may block infection by the SARS virus. We will also examine the “immunologic fitness” of these antigens by detailed analysis of antisera from the immunized mice. These studies will provide valuable information and therapeutic agents that can aid in the management of this new and emerging infectious disease. 2) 3)
=== Funded SARS Monoclonal Study === 4)
COVID Vaccine Trials and Other Studies
Many trial reports and studies on the effects of COVID-19 vaccines have been released, though many are controversial due to lack of release of raw data and methodologies used.
Vaccine Trials
Retrospective Analyses of Effects of COVID-19 Vaccines
Pharmacovigilance
The Public Health and Medical Professionals for Transparency documents include information that vaccine manufacturers wanted to keep quiet for 55 years: https://phmpt.org/pfizers-documents/
Location Studies
Prison Studies
Prison studies seem to find little measurable efficacy among the vaccines, and what little there is may be attributable to various forms of bias such as survivor bias or selection bias.
https://www.medrxiv.org/content/10.1101/2021.11.12.21265796v1
Citizen Science
- The Control Group Cooperative LTD is a citizen-funded cooperative formed for the purpose of collecting the health data of the voluntarily vaccine free as the control group to those vaccinated for COVID-19.5)
